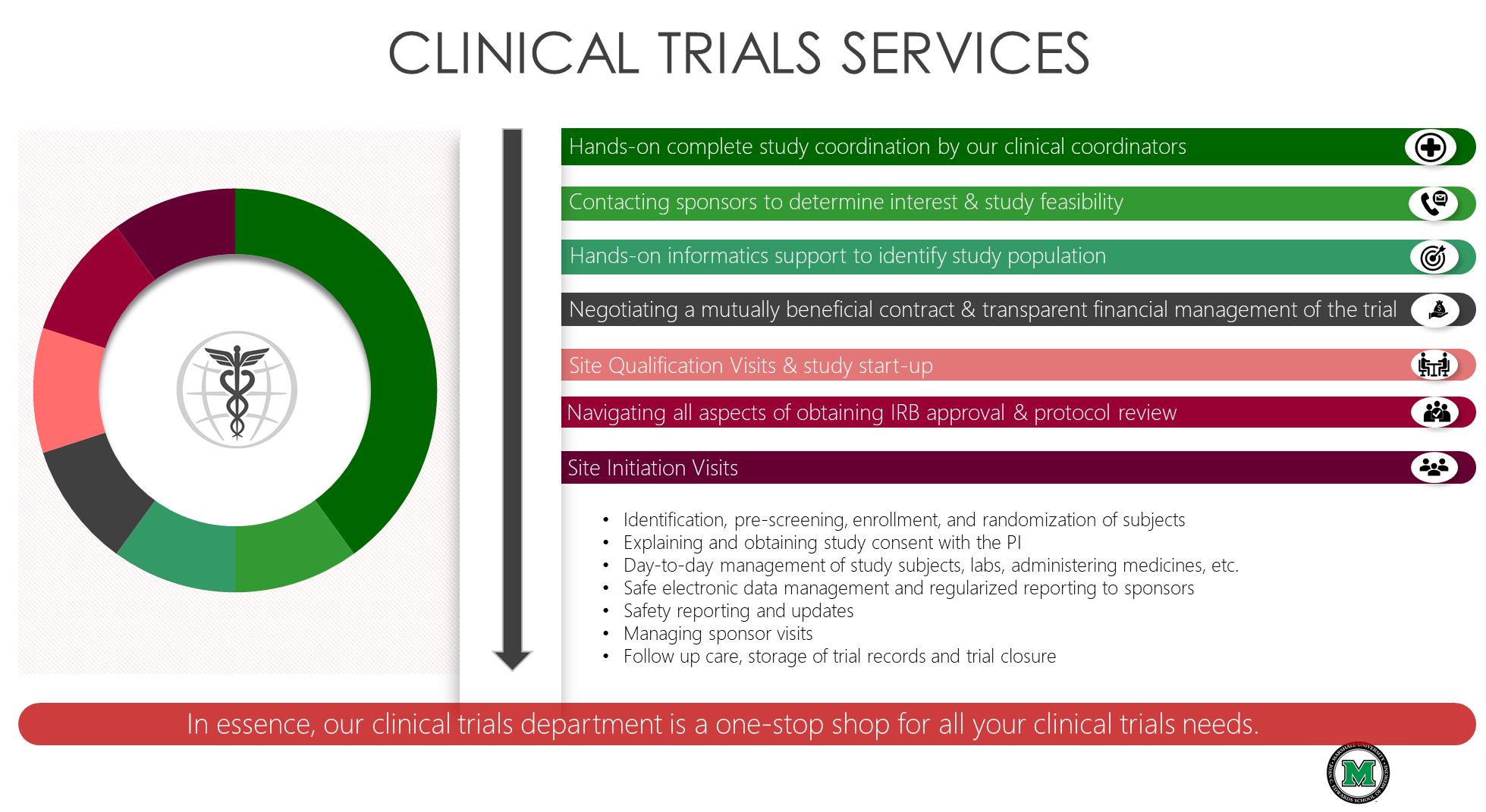
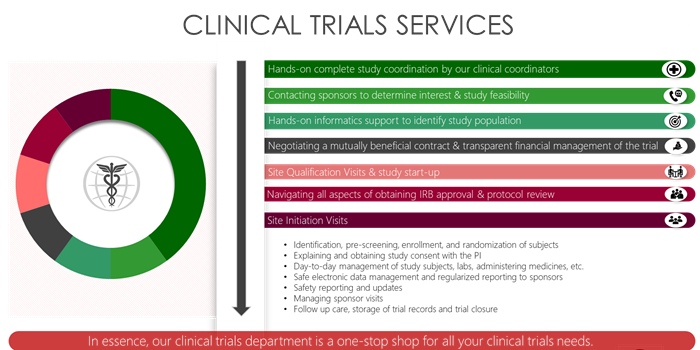
- Home |
- Research |
- Office of Research and Graduate Education |
- Departments |
- Clinical and Translational Sciences |
- Marshall Clinical Research Center
Menu 

- Current Students
- Faculty & Staff
- About
- Prospective Students
- Residents/Fellows
- GME General Info
- Incoming Residents & Fellows
- Current Residents & Fellows
- GMEC
- Important Links
- Programs
- Family & Community Health
- - Addiction Medicine Fellowship
- - Geriatric Medicine Fellowship
- - Sports Medicine Fellowship
- General Practice Residency - Dental
- Internal Medicine
- - Cardiology Fellowship
- - Endocrinology Fellowship
- - Interventional Cardiology Fellowship
- - Gastroenterology Fellowship
- - Hematology-Oncology Fellowship
- - Nephrology Fellowship
- - Nurse Practitioner Fellowship
- - Pulmonary Critical Care Fellowship
- Medicine / Pediatrics
- Neurology
- Obstetrics / Gynecology
- Orthopaedic Surgery
- Pediatrics
- - Neonatal-Perinatal Medicine Fellowship
- - Pediatric Hospital Medicine Fellowship
- Psychiatry
- - Child & Adolescent Psychiatry Fellowship
- - Geriatric Psychiatry Fellowship
- Surgery
- Quick Links
- New Innovations
- I-PASS
- Policies
- Handbook
- Research
- Departments
- Clinical Departments
- Basic Science Departments
- Divisions / Other Departments
- Animal Resources
- Forensic Science
- Health Science Library
- Human Gift Registry / Body Donation
- Clinical & Translational Sciences
- Computing / Information Technology
- Graphic Design Services
- Office of Academic Affairs
- Office of Faculty Advancement
- Office of Medical Education
- Office of Student Affairs
- Robert C. Byrd Center For Rural Health
- Administration Contacts
- Phone & Email Directory
- Alumni/Giving
- Clinical Care
- Quick Links
- Marshall Health
- Find A Doctor
- Locations
- Contact Information
- Services